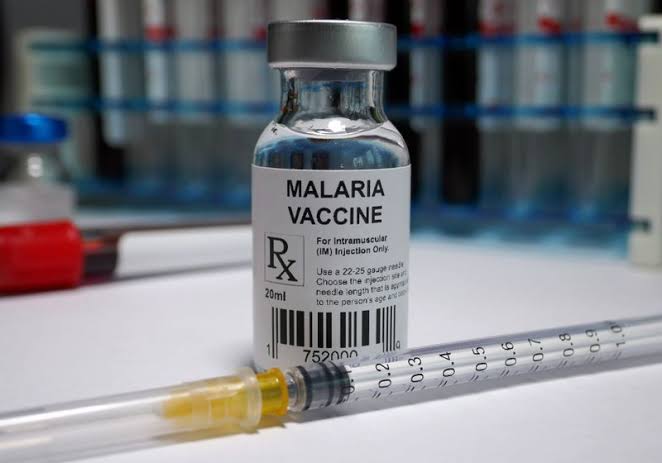
The National Agency for Food and Drug Administration and Control (NAFDAC) has granted registration approval for R21 Malaria Vaccine (Recombinant, Adjuvanted) manufactured by Serum Institute of India Pvt. Ltd. The vaccine has been developed by the University of Oxford and is indicated for the prevention of clinical malaria in children from 5 months to 36 months of age. The vaccine has already been approved by Ghana, and now Nigeria has become the second country to approve it.
The Director General of NAFDAC, Prof Mojisola Adeyeye, disclosed this on Monday at a press briefing. “NAFDAC in exercising its mandate as stipulated by its enabling law, NAFDAC Act CapN1, LFN 2004 is granting registration approval for R21 Malaria Vaccine (Recombinant, Adjuvanted) manufactured by Serum Institute of India Pvt. Ltd,” she said.
The Marketing Authorization Holder is Fidson Healthcare Ltd in line with the Agency’s Drug and Related Products Registration Regulation 2021. Adeyeye said the country expects to get at least 100,000 doses of the vaccine in donations soon before the market authorization will start making other arrangements with the National Primary Health Care Development Agency.
The R21 Malaria vaccine is an adjuvanted protein vaccine presented as a sterile solution. A dose which is 0.5ml is composed of R21 Malaria antigen 5µg and Matrix-M1 50µg as an adjuvant filled in a vial as a ready-to-use liquid formulation for intramuscular injection. The storage temperature of the vaccine is 2-8 °C.
According to Prof Adeyeye, the dossier of the vaccine was subjected to independent review at two levels. The dossier was reviewed by NAFDAC’s Vaccine Advisory Committee independently using standards of the World Health Organisation across relevant domains, in addition to the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human use guidelines, European Medicines Agency guidelines, scientific rigor on the vaccine, and the context of malaria and specifically to Nigeria and best research and manufacturing governance.
“NEVAC members reviewed all sections independently using best review practices and met physically in plenary to assess and debate the reviews by sections, raised queries, and made recommendations accordingly,” she said. “Overall, the assessment was scored as adequate – fully compliant with standards, and the report was submitted to the Director General.”
The Joint Review Committee concluded that the data on the R21 Malaria vaccine were robust and met the criteria for efficacy, safety, and quality. It was also adjudged that the vaccine’s known and potential benefits outweigh its known and potential risks, thereby supporting the manufacturer’s recommended use. “The Joint Review Committee recognised the importance of an effective malaria vaccine (with 75 per cent protection) as an additional interventional tool, as a critical need with the highest malaria burden,” she said.
“The Joint Review Committee also recommended an additional phase four clinical trial, pharmacovigilance study in-country in the implementation given the peculiarity and heterogeneous nature of malaria. The Joint Review Committee was also interested in reviewing data of the human-malaria parasite dynamics of the 25 percent not covered to understand issues bothering on non-protection that could inform further research,” she added.
According to data from the World Health Organization, four African countries accounted for just over half of all malaria deaths worldwide: Nigeria (31.3%), the Democratic Republic of the Congo (12.6%), United Republic of Tanzania (4.1%) and Niger (3.9%)., 97 per cent of the population are at risk of malaria.
The approval of the R21 malaria vaccine is a significant development in the fight against malaria, and it is expected to reduce the number of malaria-related deaths among children under five. The vaccine’s approval will also improve Nigeria’s efforts to eliminate malaria by 2025.