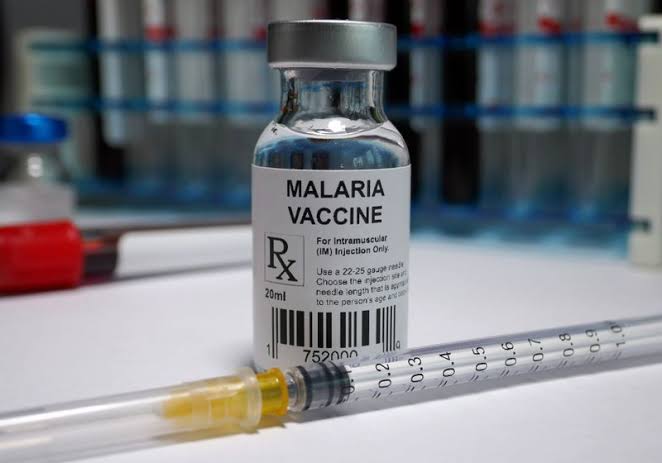
Ghana has become the first country to approve the use of a new malaria vaccine called R21. The vaccine has been developed by scientists and has been deemed a “world-changer” due to its efficacy against malaria. Malaria kills an estimated 620,000 people annually, most of whom are young children. Scientists have spent the past century attempting to develop a vaccine that can protect against the malaria parasite, and the R21 vaccine appears to be hugely effective, according to the trial data from preliminary studies in Burkina Faso, which showed that it was up to 80% effective when given as three initial doses and a booster a year later.
Despite the initial trial data, the widespread use of the vaccine is dependent on the results of a larger trial that involved almost 5,000 children. The results of this trial have not yet been formally published, but they have been shared with some government bodies in Africa and scientists. The Ghana Food and Drugs Authority has assessed the final trial data on the vaccine’s safety and effectiveness, which is not yet public, and has decided to approve its use in children aged between five months and three years old. Other African countries are also studying the data, as is the World Health Organization.
Prof Adrian Hill, director of the Jenner Institute at the University of Oxford, where the vaccine was invented, stated that African countries are declaring “we’ll decide” after being left behind in the rollout of Covid-19 vaccines during the pandemic. He further added, “We expect R21 to make a major impact on malaria mortality in children in the coming years, and in the longer term [it] will contribute to the overall final goal of malaria eradication and elimination.”
The Serum Institute of India is preparing to produce between 100-200 million doses of the vaccine per year, with a vaccine factory being constructed in Accra, Ghana. Each dose of R21 is expected to cost a couple of dollars. Adar Poonawalla, CEO of the Serum Institute, expressed his delight at Ghana being the first country to approve the vaccine and stated that the approval represents a “significant milestone in our efforts to combat malaria around the world.”
The vaccine’s approval has come as a significant relief to health experts, as it is expected to save countless lives in the fight against malaria. The vaccine’s high efficacy rate and low cost make it an accessible and effective solution for many African countries, where malaria is a significant health problem. However, the vaccine’s long-term effectiveness and safety are yet to be determined, and further trials and research are required to understand its impact fully.
Ghana’s approval of the R21 vaccine represents a significant milestone in the global fight against malaria. The vaccine’s efficacy rate and low cost make it a viable solution to tackle malaria in many African countries. However, further research and testing are required to understand the vaccine’s long-term effectiveness and safety.
Given that malaria is a leading cause of death, the approval of the R21 malaria vaccine by Ghana should serve as a wake-up call for the government to prioritize its fight against malaria. With over 300,000 deaths each year attributed to malaria, it is imperative that the country follows in Ghana’s footsteps in approving the vaccine for use in children. The government should ensure that it expedites the regulatory approval process to enable the country to take advantage of this breakthrough in malaria vaccine development, and ultimately reduce the disease’s devastating impact on its citizens.