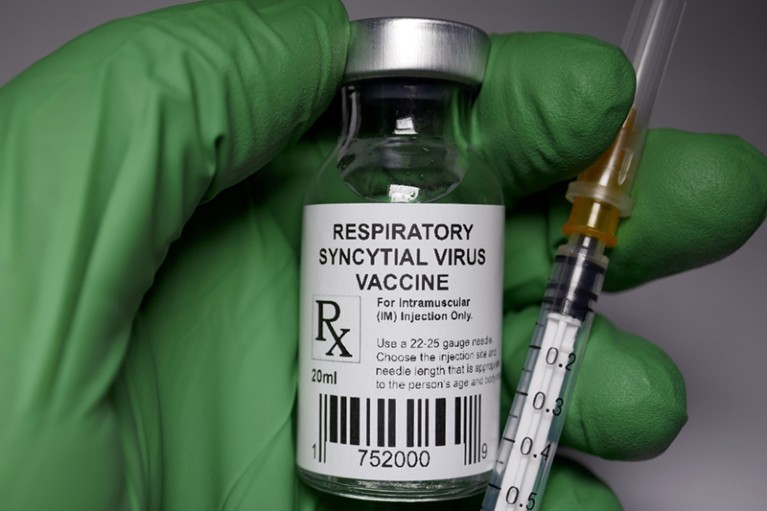
The United States of America has achieved a significant milestone by becoming the first country in the world to approve a vaccine for Respiratory Syncytial Virus (RSV), according to a statement released by the senior US Food and Drug Administration official, Peter Marks, on Wednesday.
RSV is a common respiratory virus that causes mild, cold-like symptoms in most people. However, the virus can be particularly dangerous for infants and older adults, potentially leading to pneumonia and bronchiolitis, which is an inflammation of the small airways deep inside the lungs.
The US Centers for Disease Control and Prevention has reported that RSV leads to around 60,000 to 120,000 hospitalizations and 6,000 to 10,000 deaths among adults aged 65 years and older. The strain placed on hospital systems during the last two winters has raised awareness of the disease.
Peter Marks described the approval of the RSV vaccine as “an important public health achievement” that will help to prevent a potentially life-threatening disease. The approval comes at a time when the world is battling the COVID-19 pandemic, and the development of the RSV vaccine highlights the significance of public health research and innovation in combating infectious diseases.
The approval of the RSV vaccine is a critical step towards ensuring that vulnerable populations, such as infants and older adults, are protected against the virus. With the US leading the way, it is hoped that other countries will follow suit, and this breakthrough will provide the impetus for continued investment in public health research and innovation.
While the approval of the RSV vaccine is a significant development in the fight against infectious diseases worldwide, it is particularly relevant to Nigeria, where RSV is a major cause of morbidity and mortality in children under five years of age. The disease burden of RSV highlights the need for a vaccine to protect vulnerable populations against the virus. The approval of the vaccine in the US provides hope that Nigeria and other countries facing similar challenges can soon have access to the vaccine and prevent the spread of RSV.